Active Catalysis.....................................................................................................................
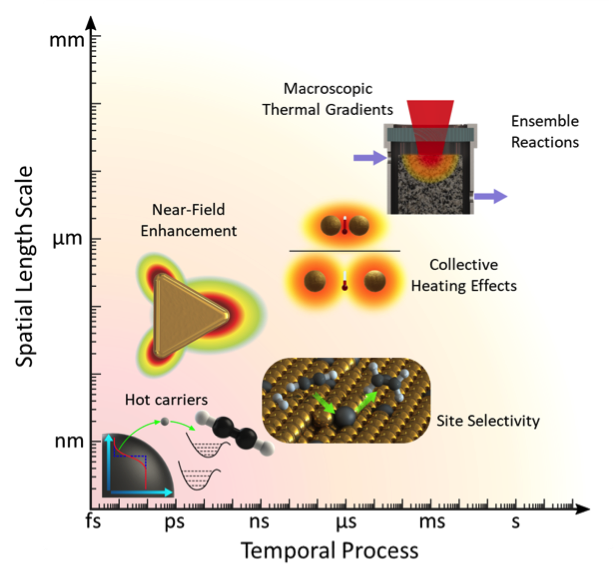
Chemical manufacturing is critical for industries spanning construction, clothing, plastics, pharmaceuticals, food, and fertilizers. These processes rely on catalysts, typically metal nanoparticles, to accelerate reaction rates, yet they remain among the most polluting and energy demanding industrial practices. More effectively controlling these chemical transformations requires bridging the length-scale between a catalyst’s atomic-scale structural features that influence dynamics and the macroscale extrinsic parameters that can be controlled (e.g., illumination, temperature, pressure). Our research focuses on enabling sustainable chemical production with atomically architected nanoparticle heterogeneous photocatalysts that precisely control optical, electronic, and molecular interactions for high-efficiency and product-selective photochemistry. Plasmons, or collective oscillations of conduction electrons within a metal, offer a solution for overcoming this size mismatch. Optical excitations of plasmon resonances in a metal nanoparticle can create nanoscopic regions of high electromagnetic field intensity that can modify electronic and molecular energy levels, enabling access to excited-state dynamics, and open new reaction pathways that are impossible to achieve under typical conditions.
The challenge our group seeks to address is understanding how these ultrafast and small optical interactions correspond to macroscopic changes in chemical activity using a multiscale approach. By revealing the multiscale photochemical processes that span Angstroms to centimeters and picoseconds to minutes, characterization will overturn the century-old empirical approach of catalyst design, and inform a new generation of sustainable, solar-driven catalysis.

Metals which are traditionally considered plasmonic, i.e. have strong plasmon resonances in the visible wavelength, such as Ag and Au, are excellent at concentrating and absorbing incoming illumination but are not very catalytically active for many reactions. On the other hand, metals commonly used for their catalytic properties, e.g. Pd for hydrogenation chemistries, have optimal electronic properties for activating certain reactant molecules at their surface but do not support plasmons in the visible and thus cannot be efficiently optically excited by solar energy. However, combining both types of metals into the same bimetallic catalyst system allows for both efficient light absorption and chemical bond activation.
Our current work explores the ability of bimetallic catalysts that incorporate both a traditionally plasmonic and catalytic metal to synergistically capture light and use that to drive chemistry with high activity, selectivity, and stability. We synthesize a variety of bimetallic nanoparticles consisting of a plasmonic and catalytic metal in well-controlled morphologies, compositions, and configurations, then characterize their catalytic and material properties on both the ensemble and single-particle level. Our catalysts have demonstrated the ability for photoexcitation to overcome the common trade-off between activity and selectivity in heterogeneous catalysis.
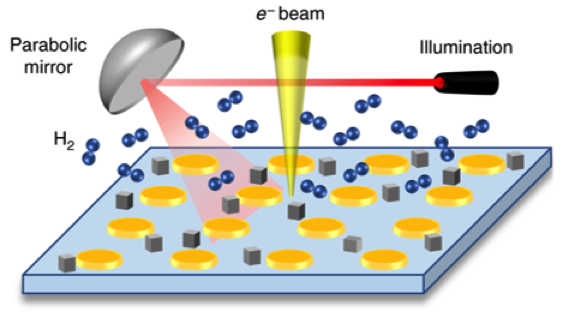
Transmission electron microscopy (TEM) techniques enable imaging and chemical and optical characterization of nanomaterials with atomic-scale resolution. Our facilities are equipped with an environmental optically-coupled transmission electron microscope (E-OTEM), wherein both light and reactive gases can be introduced into the microscope, which allows observation of gas-phase, light-driven chemical transformations in real-time.
We use E-OTEM to monitor hydrogenation reactions catalyzed by our bimetallic plasmonic and single-atom catalysts, tracking how reaction outcomes are correlated to changes in nanoscale material properties such as size, shape, composition, and crystallinity. Our findings reveal that illumination of localized plasmonic hot spots can drive site-selective chemistry that is distinct from what is favored in the dark.